AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Sodium spectral lines12/14/2023  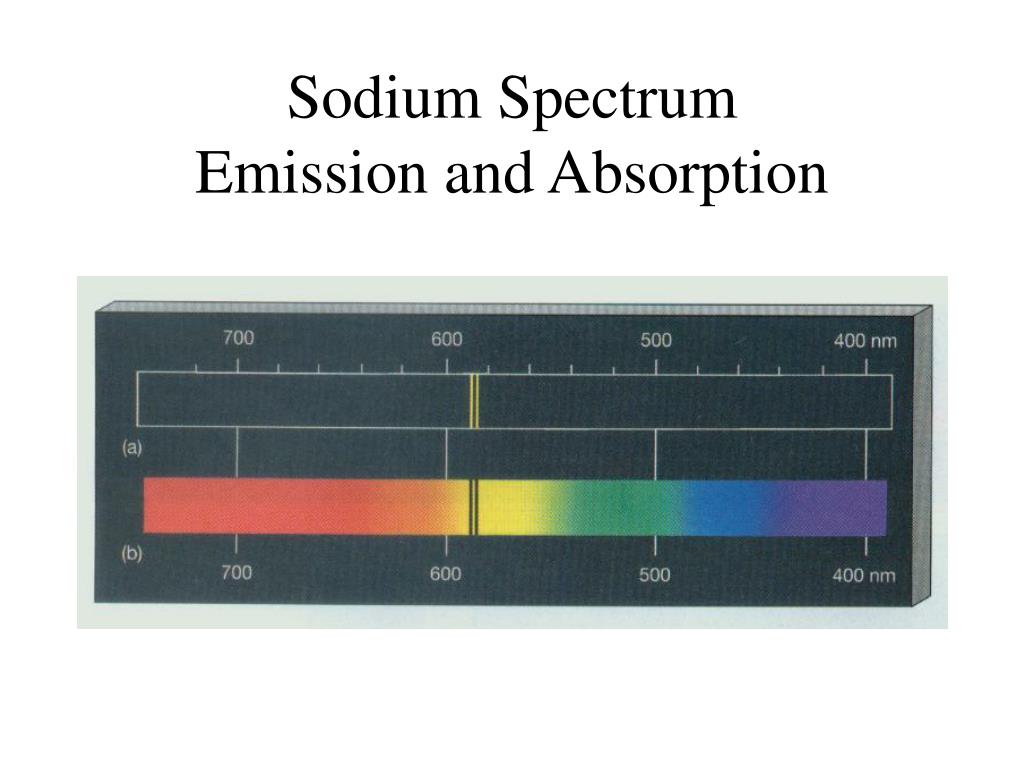 
Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron. Actually, if you just burned hydrogen and looked at its spectra, you would get the Emission Spectra and not the Absorption Spectra, and this Emission Spectra would only show the bunch of blue lines, one purple line, and one red line. When a crystalline sugar is dissolved, the rotary power of the solution slowly changes. The sodium spectrum is dominated by the bright doublet known as the Sodium D-lines at 588.9950 and 589.5924 nanometers. All the other colors shown are just part of the natural light being shown down on the element. What is the difference in energy between the two energy levels involved in the. It depends on concentration, temperature, and solvent. Sodium atoms emit a spectral line with a wavelength in the yellow, 589.6mm. 1 Rotary power is very characteristic of every sugar in solution, and is not related to its molecular weight ( Table 1 ). Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. The specific rotation D is defined using the sodium D-line at 2025 ☌. In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. The wavelengths corresponding to several transitions are shown. The origins of three of the major sodium emission spectral lines (the line at 590 nm is actually a doublet (Harris & Bertolucci, 1978)). Figure 10.57 Valence shell energy level diagram for sodium. Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. Figure 10.57 shows a portion of the energy level diagram for sodium, which consists of a series of discrete lines at wavelengths corresponding to the difference in energy between two atomic orbitals. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
